Abstract
Nivicolous myxomycete assemblages were surveyed on the northwest of the Greater Caucasian ridge in May-June 2010 and 2011 at a north facing transect between 1,700 and 2,920 m elevation of the summit Malaya Khatipara situated within the Teberda State Biosphere Reserve. Morphological characters of 396 collections representing 45 taxa (39 species, 3 varieties, and 3 forms) of myxomycetes in 8 genera and 5 families were recorded. Many (13) taxa are classified as rare (a species represents <0.5 % of all records). Only seven species were found to be widely distributed (present in 50 % or more of the 17 studied localities). To confirm the assignment of specimens to morphospecies, we obtained independently from determination 145 partial sequences of the 18S SSU rRNA gene from 35 taxa of Lamproderma, Meriderma, Physarum and Diderma, which turned out to represent 58 genotypes. Most of the taxa represented by more than one sequence had several genotypes, with an average of 1.7 genotypes per taxon. Except for three taxonomically difficult groups of species, partial SSU sequences did well correspond with the respective morphospecies and where similar or identical to sequences of specimens from the European Alps, making this marker a good candidate for barcoding in myxomycetes. Species richness and diversity increased from subalpine crooked-stem birch forests (23 species, 2 varieties, H′ = 2.8, E = 0.88, D = 0.08) to alpine dwarf shrub communities (34 species and 2 varieties, 2 forms, H′ = 3.2, E = 0.89, D = 0.05) but decreased again for alpine meadows (27 species and 2 varieties, 2 forms, H′ = 3.1, E = 0.91, D = 0.06). Species richness and alpha-diversity reached maximum values for ground litter, whereas leaves and stems of living shrubs above ground harboured a more depauperate myxomycete assemblage.











Similar content being viewed by others
References
Anonymous (2012) NBS/ISCC color system. Original and improved 267 color centroids. Washington: Inter-Society Color Council. National Bureau of Standards. http://tx4.us/nbs-iscc.htm. Accessed 28 July 2012
Chao A, Chazdon RL, Colwell RK, Shen TJ (2005) A new statistical approach for assessing similarity of species compostion with incidence and abundance data. Ecol Lett 8:148–159
Chao A, Chazdon RL, Colwell RK, Shen TJ (2006) Abundance-based similarity indices and their estimation when there are unseen species in samples. Biometrics 62:361–371
Colwell RK (2009) EstimateS: statistical estimation of species richness and shared species from samples. Version 8.2. User’s guide and application. http://purl.oclc.org/estimates. Accessed 12 April 2012
Colwell RK, Chao A, Gotelli NJ, Lin S-Y, Mao CX, Chazdon RL, Longino JT (2012) Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. J Plant Ecol 5(1):3–21
Elumeeva TG, Salpagarov AD, Onipchenko VG (2007) Dynamics of temperature and precipitation in Karachaevo-Cherkesia during the second part of XX century. Trans Teberda Reserv 27:20–29
Fiore-Donno AM, Meyer M, Baldauf SL, Pawlowski J (2008) Evolution of dark-spored myxomycetes (slime-molds): molecules versus morphology. Mol Phylogenet Evol 46(3):878–889
Fiore-Donno AM, Novozhilov YK, Meyer M, Schnittler M (2011) Genetic structure of two protist species (Myxogastria, Amoebozoa) suggests asexual reproduction in sexual amoebae. PLoS One 6(8):e22872. doi:22810.21371/journal.pone.0022872
Fiore-Donno AM, Kamono A, Meyer M, Schnittler M, Fukui M, Cavalier-Smith T (2012) 18S rDNA Phylogeny of Lamproderma and allied genera (Stemonitales, Myxomycetes, Amoebozoa). PLoS One 7(4):e35359. doi:10.1371/journal.pone.0035359
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp 41:95–98
Kamono A, Matsumoto J, Kojima H, Fukui M (2009a) Characterization of myxomycete communities in soil by reverse transcription polymerase chain reaction (RT-PCR)-based method. Soil Biol Biochem 41:1324–1330
Kamono A, Kojima H, Matsumoto J, Kawamura K, Fukui M (2009b) Airborne myxomycete spores: detection using molecular techniques. Naturwissenschaften 96(1):147–151
Karlsen A (1943) Studies on myxomycetes II. The myxomycete flora of Hardangen. Bergens Museums Naturvitenskapelig Rekke 4:1–34
Kirk P (2012) Authors of fungal names. http://wwwindexfungorumorg/authorsoffungalnameshtm. Accessed 15 April 2012
Kowalski DT (1971) The genus Lepidoderma. Mycologia 63:490–516
Kuhnt A (2011) Lamproderma lycopodiicola und L. nordica (Myxomycetes, Stemonitales), zwei neue nivicole Arten. Z Mykologie 77:71–88
Lado C (2004) Nivicolous myxomycetes of the Iberian Peninsula: considerations on species richness and ecological requirements. Syst Geogr Plant 74:143–157
Lado C (2005–2012) An on-line nomenclatural information system of Eumycetozoa. http://www.nomen.eumycetozoa.com accessed. Accessed 10 February 2012
Lado C, Ronikier A (2008) Nivicolous myxomycetes from the Pyrenees: notes on taxonomy and species diversity. Part 1. Physarales and Trichiales. Nova Hedwigia 87(3–4):337–360
Lado C, Ronikier A (2009) Nivicolous myxomycetes from the Pyrenees: notes on the taxonomy and species diversity. Part 2. Stemonitales. Nova Hedwigia 89(1–2):131–145
Lado C, Ronikier A, Ronikier M, Drozdowicz A (2005) Nivicolous myxomycetes from the Sierra de Gredos (central Spain). Nova Hedwigia 81(3–4):371–394
Magurran AE (2004) Measuring biological diversity. Blackwell Publishing, Malden
Makarov MI, Onipchenko VG, Malysheva TI, Salpagarov AD (2007) Temporal dynamics of soil temperature and climatic parameters of alpine ecosystems in the scientific station “Malaya Khatipara”. Trans Teberda Reserve 27:30–41
Meylan C (1914) Remarques sur quelques espèces nivales de myxomycètes. Bull Soc Vaud Sci Nat 50(182):237–244
Mitchel DH, Chapman SW, Farr ML (1986) Notes on Colorado fungi V: Physarum alpestre, a new species. Mycologia 78:66–69
Moreno G, Sanchez A, Castillo A, Singer H, Illana C (2003) Nivicolous myxomycetes from the Sierra Nevada National Park (Spain). Mycotaxon 87:223–242
Moreno G, Singer H, Illana C (2004) A taxonomic review on the nivicolous myxomycete species described by Kowalski. II. Physarales and trichiales. Oesterr Z Pilzk 13:61–73
Moreno G, Singer H, Sanchez A, Illana C (2005) A critical study of some Stemonitales of North American herbaria and comparison with European nivicolous collections. Bol Soc Micol Madrid 28:21–41
Müller H (2002) Beitrag zur Kenntnis und Verbreitung nivicoler Myxomyceten im Thüringer Wald. Z Mykologie 68(2):199–208
Novozhilov YK (1986) Nivicolous myxomycetes of the Leningrad region. Novosti Sistematiki Nizshikh Rastenii 23:146–149 (in Russian)
Novozhilov YK, Schnittler M (1997) Nivicole myxomycetes of the Khibine mountains (Kola peninsula). Nord J Bot 16:549–561
Onipchenko VG (ed) (2004) Alpine ecosystems in the Northwest Caucasus. Kluwer Academic Publishers, Dordrecht, Boston; London
Onipchenko VG (2007) Syntaxonomy of alpine vegetation in the Teberda reserve: syntaxa prodromus and diagnostic species. Trans Teberda Reserv 27:75–82
Poulain M, Meyer M, Bozonnet J (2011) Les Myxomycètes, vol 1–2. Fédération mycologique et botanique Dauphiné-Savoie
Ronikier A, Ronikier M (2009) How ‘alpine’ are nivicolous myxomycetes? A worldwide assessment of altitudinal distribution. Mycologia 101(1):1–16
Ronikier A, Ronikier M, Drozdowicz A (2008) Diversity of nivicolous myxomycetes in the Gorce mountains — a low-elevation massif of the western Carpathians. Mycotaxon 103:337–352
Ronikier A, Lado C, Meyer M, de Basanta DW (2010) Two new species of nivicolous Lamproderma (myxomycetes) from the mountains of Europe and America. Mycologia 102(3):718–728
Schnittler M (2001) Ecology of Myxomycetes of a winter-cold desert in western Kazakhstan. Mycologia 93(4):653–669
Schnittler M, Novozhilov YK (1999) Lepidoderma crustaceum, a nivicolous myxomycete, found on the island of Crete. Mycotaxon 71:387–392
Singer H, Moreno G, Illana C (2005) Mountainous and nivicolous myxomycetes described by Charles Meylan. A SEM-study. Oesterr Z Pilzk 14:11–29
Stephenson S (2001) Collecting myxomycetes and fungi in Snowbank habitats of New Zealand. Inoculum Suppl Mycol 52(3):1–2
Stephenson SL (2011) From morphological to molecular: studies of myxomycetes since the publication of the Martin and Alexopoulos (1969) monograph. Fungal Divers 50(1):21–34
Stephenson SL, Shadwick JDL (2009) Nivicolous myxomycetes from alpine areas of south-eastern Australia. Aust J Bot 57(2):116–122
Stephenson SL, Kalyanasundaram I, Lakhanpal TN (1993) A comparative biogeographical study of myxomycetes in the mid-Appalachians of eastern North America and two regions of India. J Biogeogr 20:645–657
Stephenson SL, Fiore-Donno AM, Schnittler M (2011) Myxomycetes in soil. Soil Biol Biochem 43:2237–2242
Tamayama M (2000) Nivicolous taxa of the myxomycetes in Japan. Stapfia 73:121–129
Unterseher M, Schnittler M, Dormann C, Sickert A (2008) Application of species richness estimators for the assessment of fungal diversity. FEMS Microbiol Lett 282:205–213
Wuyts J, Van de Peer Y, Winkelmans T, De Wachter R (2002) The European database on small subunit ribosomal RNA. Nucleic Acids Res 30:183–185
Yajima Y, Nishikawa T, Yamamoto A (2006) Studies on the myxomycetes of Hokkaido, Japan (II). Nivicolous species of Mt. Asahidake in Mts. Taisetsu. Rep Taisetsuzan Inst Sci 40:53–57
Acknowledgments
We gratefully acknowledge logistical help provided by A.N. Bok (Teberda State Biosphere Reserve) and V.N. Khramtzov (Komarov Botanical Institute RAS) as well as technical support of SEM by L.A. Kartzeva (Komarov Botanical Institute). For help with sequencing we are indebted to A. Klahr, for help with alignments to A.M. Fiore-Donno (both Greifswald). Travel and laboratory work were supported by the grant RFBR 10–04–00536a to the first author as well as a scientific program “Bioraznoobrazie” from the Russian Academy of Sciences. Travel for the second author was supported by grants from Greifswald University, sequencing in part by a grant from the Deutsche Forschungsgemeinschaft (SCHN1080/2-1). We are grateful to David Mitchell for comments and linguistic correction and Marianne Meyer for help with identification of specimens.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Supplement S1
List of specimens found in this survey, presented as a data base in Microsoft Excel. 2003. (XLS 214 kb)
Supplement S2
Alignment of the partial SSU sequences for Stemonitales (including the genus Diachea which can be seen as a member of the Physarales as well) in fasta format; including matching sequences drawn from GenBank as well as unpublished comparison material. To avoid long-branch attraction in the resulting trees, Stemonitopsis spp. and Lamproderma arcyrionema, showing extremely divergent partial SSU sequences, have been omitted. (FAS 217 kb)
Supplement S3
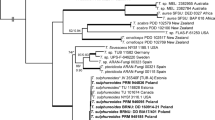
Maximum likelihood phylogenetic tree of the partial SSU sequences for Stemonitales, reconstructed with IQTree software (http://www.cibiv.at/software/iqpnni/) under the GTR + I + G model, in Newick format, readable with free software like FigTree v.1.3.1. (http://tree.bio.ed.ac.uk/software/figtree/). Although the topology of the tree basically corresponds to a tree constructed using complete SSU sequences published by Fiore-Donno et al. (2012), it should be noted that the short partial SSU sequences are not suitable to derive correct phylogenetic relationships between species. (PDF 9 kb)
Supplement S4
Alignment of the partial SSU sequences for Physarales (including the genus Diachea) in fasta format; including matching sequences drawn from GenBank as well as unpublished comparison material. (FAS 114 kb)
Supplement S5
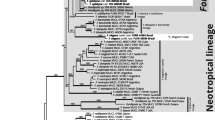
Maximum likelihood phylogenetic tree of the partial SSU sequences for Physarales, reconstructed with IQTree software under the GTR + I + G model, in Newick format. (PDF 8360 kb)
Rights and permissions
About this article
Cite this article
Novozhilov, Y.K., Schnittler, M., Erastova, D.A. et al. Diversity of nivicolous myxomycetes of the Teberda State Biosphere Reserve (Northwestern Caucasus, Russia). Fungal Diversity 59, 109–130 (2013). https://doi.org/10.1007/s13225-012-0199-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13225-012-0199-0