Abstract
The response of species to the environment is scale-dependent and the spatial scale at which this relationships are measured may affect conservation recommendations. Saproxylic beetles depend on decaying- and deadwood which occur in lower quantities in managed compared to natural forests. Most studies have investigated the habitat selection of saproxylic beetles at the stand scale, however depending on the species mobility, the amounts and distribution of forest attributes across the landscape may be equally important, and thus crucial to frame quantitative conservation targets. To address this gap, we evaluated the influence of environmental variables, derived from remote sensing across multiple spatial scales (50, 100, 250, 500 and 1000 m radius), on saproxylic beetles habitat selection. Focusing on four mobile and four flightless species, we hypothesized that mobile species respond to habitat variables at broader scales compared to flightless species, and that variables describing forest structure explain species presence better at smaller scales than variables describing other landscape features. Forest structure variables explained around 40% of the habitat selection, followed by variables describing forest type, topography and climate. Contrary to our expectations, mobile species responded to variables at smaller scales than flightless species. Saproxylic beetle species therefore respond to the availability of habitat features at spatial scales that are inversely related to their dispersal capacities, suggesting that less mobile species require larger areas with suitable habitat characteristics while mobile species can also make use of small, distributed patches with locally concentrated habitat features.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forested areas cover about one-third of the terrestrial earth surface (FAO 2010) and provide habitats for many species. However, this biome is strongly altered by the need to reconcile multiple anthropogenic and ecological demands resulting in multifunctional forest management (Lindenmayer and Franklin 2002). In densely populated regions such as Central Europe, close-to nature forestry is the predominant management regime, targeting economical goals while attempting to conserve biodiversity (Bauhus et al. 2013). Close-to nature forestry focuses on selective logging and natural regeneration. By doing so, it promotes vertical structural complexity at the stand-scale, however it also results in a structural homogenization at the landscape scale (Bauhus et al. 2013) for example by the lack of disturbances and the resulting early successional stages and light availability (Gustafsson et al. 2020). In addition, as harvesting takes place when trees reach maturity, these stands lack late successional stages with senescent trees and high amounts and diversity of deadwood. This large-scale homogenization and impoverishment of key structures has led to a decrease in forest species richness (Paillet et al. 2010), which becomes particularly evident in the ongoing insect decline, even in forest ecosystems (Seibold et al. 2019).
Forest specialist species like saproxylic beetles represent a group that is particularly sensitive to current forest management practices (Schmidl and Bussler 2004), which is evident in the high proportion (27%) of threatened species in Germany (Seibold et al. 2015). As saproxylic beetles depend on deadwood in at least one stage of their life cycle (Speight 1989), they are frequently used as indicators for deadwood amount and diversity, which is associated with forest naturalness and structural complexity (Lachat et al. 2012; Gao et al. 2015). Although the amount (Müller et al. 2007) and the diversity of deadwood (Seibold et al. 2016) has been identified as the most influential structural habitat requisite, saproxylic beetles greatly vary in their habitat requirements and different forest structural elements such as decay stage of deadwood or light availability (Müller et al. 2015; Seibold et al. 2016).
Environmental conditions and forest structures at the stand scale are important for saproxylic beetle habitat selection (e.g. Hjältén et al. 2012; Kraut et al. 2016; Seibold et al. 2016). However, the drivers of species presence are not only operating at the stand scale, but as well at the landscape scale (Seibold et al. 2019). Selecting the right spatial scale is important, as ecological processes and patterns can only be detected, when being addressed at the spatial scale they occur (Levin 1992). Spatial scale has two components, namely the spatial grain and spatial extent. Spatial grain (in the following used synonymously with spatial scale) is defined as the resolution of the analysis, while spatial extent defines the area covered by the study.
For beetles, various spatial scales have been used to study species-environment relationships, with single-scale models ranging between 20 m (Dittrich et al. 2019), 60 m (Crawford and Hoagland 2010), 100 m (Judas et al. 2002; Kärvemo et al. 2014; Della Rocca et al. 2017), 1 km (Hof and Svahlin 2016; Silva et al. 2016; Brunetti et al. 2019) or even 10 km resolution for Great Britain (Eyre et al. 2005; Jiménez-Valverde et al. 2007). However, little is known about the optimal spatial scale at which the selection of particular resources or habitat structures actually takes place (Sverdrup-Thygeson et al. 2014). A few studies have evaluated the optimum spatial scale for different habitat variables for predicting saproxylic beetles presence in forests (Økland et al. 1996; Holland et al. 2004; Bergman et al. 2012; Jacobsen et al. 2015). The evaluated optimal scales ranged between 20 and 2000 m for forest cover (Holland et al. 2004), 50-5000 m for substrate density (Bergman et al. 2012), 2000–3000 m for forest age and volume (Jacobsen et al. 2015) and up to 1000–4000 m for various other habitat characteristics. The spatial scale at which habitat selection occurs is generally related to the species’ perception of the landscape and thus varies greatly among taxonomic groups (Turner et al. 2019). One trait that may affect scale selection is the species mobility. Species with the capacity to fly may have larger ranges and a higher capacity to move through unsuitable habitat (Steffan-Dewenter et al. 2002; Chust et al. 2004; Percel et al. 2019).
There is growing evidence, that habitat models including variables at multiple scales (multi-scale models) perform better than models using all predictors at the same scale (single-scale models) (Bergman et al. 2012). The advancement in the field of remote sensing has led to area wide datasets with different spatial resolutions and facilitated studies about environment-habitat relationships at multiple scales (He et al. 2015; Reddy et al. 2021).
In order to explore the effects of spatial scales on the occurrence of eight saproxylic beetle species we applied species distribution models. In the following we will use the term habitat selection (sensu McGarigal et al. 2016) when referring to the performance of environmental variables in explaining species presences’. We used high-resolution remote sensing data measured within incrementally increasing radii to predict the presence of species with different dispersal abilities (mobility), four flying (in the following termed as “mobile”) and four flightless species. Evaluating the spatial scales at which different variables performed best, we hypothesized, that (i) flightless species respond at smaller spatial scales compared to mobile species (Gehring and Swihart 2003), and that (ii) variables representing forest structure and resources are responding at smaller spatial scales compared to variables describing general conditions such as topography and forest type in all species. In addition, we (iii) hypothesize a better performance of multi-scale models compared to single-scale models for area-wide predictions of species distributions. With the identification of important habitat requisites and the corresponding scales at which they support the presence of species belonging to a conservation-relevant taxonomic group, our results will provide quantitative target values for large-scale conservation strategies enhancing structural complexity in managed temperate forests.
Materials and methods
Study area
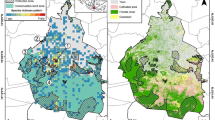
The study was conducted in the Black Forest in southwestern Germany (see Fig. 1). It is a lower mountain range with altitudes ranging from 120 to 1493 m.a.s.l. and 75% of the area is covered by forests, with Picea abies (42.8%), Abies alba (18.5%) and Fagus sylvatica (15.3%) being the dominant tree species. The forests are managed with a close-to-nature management strategy leading to continuous-cover forests, see Storch et al. (2020) for more details. Within the study area of 7167 km2 in size, beetles were collected at 180 one hectare plots, that had been established in two large-scale research programs run by the University of Freiburg (for details see: Storch et al. (2020), and the Forest Research Institute of Baden-Württemberg (FVA, for details see Eckerter et al. (2022). Plots were selected to represent a gradient of standing deadwood and amount of forest surrounding these plots and restricted to locations with < 35° slope, elevation of > 400 m.a.s.l. and stand age > 60 years (Storch et al. 2020). The maximum possible distance between the surveyed plots was 138 km.
The study area (black) is located in the Black Forest in the state of Baden-Württemberg (grey) in south west of Germany (white, A). Beetles were collected at 180 study plots (B) with flight interception traps for flying species only (blue triangles), and a combination of pitfall traps or hand collection for flightless species and flight interception traps (pink triangles). Forest above 400 m.a.s.l. is highlighted in green, grey represents forest below that threshold and non-forested area
Collection and selection of model species
We selected four flying species (mobile), namely Ampedus balteatus, Salpingus ruficollus, Hedobia imperialis and Ostoma ferruginea as they represented different families and preference for different forest types, from coniferous to broadleaved-mixed dominated forests. As flightless species we selected Pterostichus pumilio (non-saproxylic), Adexius scrobipennis, Acalles micros and Echinodera hypocrita. An overview of all species including mobility, forest type preference and their ecological guild is given in Table 1.
Species presence data were obtained using different methods: Flying beetles were sampled with two flight interception traps per plot over a growing season as described in Knuff et al. (2019). Flightless beetles were sampled by hand (leave litter sifting) next to deadwood and with three pitfall traps over one month or more. More details about sampling locations, methods and sampling effort can be retrieved from table S1. As we used a presence-only model for the analyses, only one presence location per species was retained per plot and allocated to the plot center. Moreover, we reduced the dataset allowing a minimal distance of 400 m between presence locations using the function thin from the SDMtune R-package (Vignali et al. 2020). The number of presence locations per species varied and are listed below and in Table 2.
Environmental variables
We used environmental variables describing climate, topography, forest type and forest structure (Table 3). Forest structure variables were derived mainly from vegetation height models and orthophotos based on stereo aerial photographs of a ground resolution of 0.2 × 0.2 m, dated from 2015 to 2017 and supplied by the State Office for Geoinformation and Land Development Baden-Württemberg (2018). Digital orthophotos and 3D photogrammetric point clouds were produced from aerial imagery using the image-matching software SURE of nFrames (nFRAMES GmbH Stuttgart: https://www.nframes.com; Rothermel et al. 2012). The latter served then as input to calculate vegetation height models with a resolution of 1 × 1 m as described in Schumacher et al. (2019) and Ganz et al. (2020), which was then used to calculate forest height and forest height heterogeneity. Standing deadwood (Zielewska-Büttner et al. 2020), gaps and open forest (Zielewska-Büttner et al. 2016) were also calculated based on aerial imagery products (for details see table S2).
Sentinel 2 data (downloaded from the Copernicus Open Access Hub https://scihub.copernicus.eu/) originating from the years 2016–2018 were used for modelling tree volume according to Schumacher et al. (2019) as well as forest type. For forest type classification support vector machine regression models were applied using optical remote sensing data. Pure broadleaved forests were classified as having more than 80% estimated proportion of trees per pixel, mixed broadleaved 20–100%, mixed 20–80%, coniferous mixed 0–80% and pure coniferous less than 20% deciduous trees. Most variables were developed at the Forest Research Institute (FVA) of Baden-Württemberg and provided through the MoBiTools project (https://www.fva-bw.de/top-meta-navigation/fachabteilungen/biometrie-informatik/mobitools). Tree cover density was retrieved from Copernicus Land Monitoring Service (https://land.copernicus.eu) for the year 2017.
Topographic variables were calculated using a digital elevation model with an original resolution of 1 × 1 m, as supplied by the LGL (2018). Slope, eastness, northness and roughness were generated using the terrain function from the raster R-package (Hijmans et al. 2022). The topographic position index were calculated with the spatialEco R-package (Evans et al. 2021). Climatic variables had an original resolution of 250 m and were provided by the Department of Physical Geography at the University of Hamburg (Dietrich et al. 2019). The variables represent summarized annual values for the period 1991 to 2018.
Variable preparation
All environmental variables were resampled to the same resolution (20 × 20 m) using bilinear interpolation for continuous, and the nearest neighbor method for discrete raster values and cropped to the extent of the study area. In addition, variables were masked to areas with an elevation > 400 m.a.s.l. and defined as forest, including temporarily treeless forest areas, e.g. windthrow areas and gaps (Ganz et al. 2020).
We calculated average values within circular moving windows with radii of 50, 100, 250, 500 and 1000 m for all of the variables, except those representing climate, which were kept at their original resolution of 250 m. We did not exceed the scale of 1000 m as we did not assume that beetles’ respond to landscape elements operates beyond this scale and to limit the maximum overlap between the radii of neighboring presence plots.
Univariate species distribution models and scale selection
To model species habitat selection we used a presence only approach (Maxent, Phillips et al. 2006) as sampling effort per plot was not standardized. The Maxent algorithm is known for its high predictive accuracy, even with small sample sizes (Elith et al. 2006; Guisan et al. 2007; Turner et al. 2019). The presence locations were contrasted against 10,000 random background locations, that were selected with the RandomPoints function in the R-package dismo (Hijmans et al. 2020).
Model calibration, tuning and evaluation was performed using the R-package SDMtune (Vignali et al. 2020). First, the presence data for each species were split in test (30%) and training (70%) datasets. For each species univariate SDM’s were then trained with each variable at each scale separately. In order to account for slight differences between evaluation metrics and obtain more stable results, we used three different metrics to evaluate the univariate models: Akaike’s information criterion corrected for small sample sizes (AICc) (Burnham and Anderson 2002), as well as the area under the receiver operating characteristic (ROC) curve (AUC) (Fielding and Bell 1997) and the true skill statistic (TSS) (Allouche et al. 2006), both applied to test the model’s fit on the training dataset (”train”) and the predictive accuracy on the test dataset (“test”). Scale selection was then performed using an ensemble metric approach, based on the five different evaluation metrics calculated on the univariate models for each variable and scale (table S3). For this purpose, for each metric the difference between the best and least performing scale of the same variable was scaled to a value between 0 and 1, and the relative score for each scale calculated. The values for AICc were reversed (as the lowest value represents the best performing models) and the resulting scores of the different metrics summed up to an ensemble value. The scale with the lowest ensemble value was selected as the “best scale”. Finally, we retained only variables which explained species presence better than random at their best-performing scale, i.e. when the respective model had an AUC test of more than 0.5.
We performed a Wilcox test to investigate the differences in scale selection between flightless and mobile species and a Kruskal-Wallis test for testing differences between scales at which the different variable types were selected. In addition, we used a generalized mixed effect model (nlme R-package; Pinheiro et al. 2020) to test if the ensemble values, as a measure of consistency in scale selection, differed in relation to variable type and species mobility.
Multivariate species distribution models/habitat selection
The retained variables at their “best scale” were included in a multivariate model. Of pairs or groups of correlated variables (Spearman’s R > |0.7|) only the one was retained that performed best (based on the AICc) in univariate models. For each species, multivariate models were then trained with the feature class combinations depending on the number of presence locations as recommended by Phillips and Dudík (2008): For Acalles micros (9 presence points) only linear features (“l”) were used, for Adexius scrobipennis (10), O. ferruginea (17), E. hypocrita (26), P. pumilio (39), Ampedus balteatus (63) and H. imperialis (67) linear and quadratic features (“lq”) were tested, and for S. ruficollis (123) product features (“lqp”) were additionally allowed. In order to optimize model parsimony, variables were reduced using the reduceVar function (R-package SDMtune) for each species with more than 20 presence locations. This function stepwise removes the least performing variables until all variables have a permutation importance > 7% (based on 10 permutations). To avoid overparameterization of models for species with 10–20 presence points (i.e. O. ferruginea and Adexius scrobipennis), we reduced the predictor variable set to two variables (Harrell et al. 1996) testing all possible variable combinations. For Acalles micros with only nine presence locations we allowed only one variable.
For the best models and the second-best candidate models (AICc difference < 2) hyperparameters were tuned using the optimizeModel function. This method relies on a genetic algorithm to find the optimal hyperparameter combination (for details see: Vignali et al. (2020). Regularization multiplier values between 0.1 and 5 with an increment of 0.05 were tested, and possible feature class combinations allowed as described above. Among the candidate models the model with the lowest AICc was selected as final model.
In order to compare multi-scale with single-grain models, we repeated the workflow described above, but selected all variables at the same scale (50, 100, 250, 500 and 1000 m radius). The resulting five models were then evaluated and compared with the multi-scale model using the AICc and AUC.
Results
Scale selection
The “best scale” at which each variable was selected by each species is reported in Table 4. Both mobility and variable types affected scale selection (see Fig. 2). Mobile species responded to variables at smaller scales (median = 50) than flightless species (median = 250; Wilcoxon rank sum test: p = 0.003). Forest structure variables were selected at smaller scales (median = 100) than forest type (median = 250) and topographic variables (median = 250; Kruksal-Wallis chi squared = 10.08, p = 0.006). Neither did the ensemble value significantly differ between flightless and mobile species (p = 0.152), nor between forest structure and forest type (0.414) but for the topographic variables (p = 0.023), with the latter correlation being characterized by higher ensemble values, i.e. lower consistency in scale selection.
“Optimal scale”, as identified by an ensemble evaluation metric, at which saproxylic beetles responded to variables (N = 8), in relation to the variable type (with STRUC = forest structure, TOPO = topography, TYPE = forest type) (A) and the species mobility (B). Ensemble metric values of the responding “best scales” showing the uncertainty in scale selection, with higher values reflecting higher variability between the optimal scales obtained with different evaluation metrics, in relation to variable type (C) and species mobility (D)
Habitat selection
The multivariate models showed a moderate (O. ferruginea AUC = 0.77) to excellent (Adexius scrobipennis AUC = 0.95) performance, with the flightless species presences being predicted with an overall higher accuracy (Table 2). Projected probabilities of species occurrence are shown in Fig. 3. Over all species, forest structure variables were contributing most to the final models with on average 40.25%, closely followed by forest type 38.32%. Topographic and climatic variables explained 13.69% and 7.72%, respectively.
Predicted probability of species occurrence across the study area ranging from yellow (low probability) to blue (high probability). Mobile species are shown in the upper, flightless species in the lower panel. Red dots indicate the presence locations and areas below 400 m.a.s.l. and non-forested area are indicated in white
The presence of Ampedus balteatus was positively influenced by a high proportion of coniferous forest at a 50 m scale and of gaps at a 1000 m scale, low humidity and to a lesser extent by tree cover at a 50 m scale. Additionally, east facing slopes (1000 m) and positive topographic position index values (100 m), i.e. exposed positions, were increasing the probability of occurrence. Salpingus ruficollis occurred at high elevations (50 m scale), flat terrain (i.e. low roughness) (50 m) and north facing slopes (1000 m). It preferred coniferous mixed forest (100 m) embedded in broadleaved forest in a wider surrounding (1000 m), with low amounts of gaps and a high tree volume, both at the 50 m scale. Hedobia imperialis responded strongest to forest height, preferring high forests at a 50 m scale and high tree density at a 100 m scale. High elevation (50 m scale) and western facing slopes (500 m) had a positive effect on its occurrence. Low proportions of coniferous forest cover at a 1000 m scale and high mixed cover at 250 m scale additionally influenced their presence. The presence of O. ferruginea depended on a high tree cover and high amounts of standing deadwood, both at the 50 m scale. The flightless beetle P. pumilio preferred low temperatures and low amounts of gaps and open forests at a 50 m scale, intermediate to high amounts of standing deadwood at a 100 m scale and positive topographic position index values at the 50 m scale. The presence of Adexius scrobipennis was best explained by broadleaved-mixed forest (250 m) and forest height (50 m), both positively affecting its presence. Broadleaved-mixed forest best explained the presence of Acalles micros at 250 m scale. Intermediate amounts of mixed forest (500 m), greater forests heights (100 m) and a high elevation (50 m) were best predicting the presence of E. hypocrita. Response plots for each of the variables are depicted in Fig. S1.
Single versus multi-scale models
Multi-scale models performed generally better than the corresponding single-scale models, although some single-scale models outperformed the multi-scale models when the AUC was used as evaluation metric (see Fig. 4). The multi-scale models for Ampedus balteatus, S. ruficollis, H. imperialis and E. hypocrita predicted better than any of the single-scale models. However, the single-scale models for Acalles micros, Adexius scrobipennis, O. ferruginea and P. pumilio at the scales 100, 50 and 100, 250 and 50, respectively had slightly higher AUC values. However, when model quality was assessed using the AICc, all multi-scale models performed better or just as good (delta AICc < 2) as the single-scale models (see table S4).
Discussion
We evaluated scale-dependent habitat selection of saproxylic beetles, comparing species with different dispersal capacities, i.e. highly mobile vs. flightless species. The selected species showed distinct requirements with regard to forest structure, forest type, topography and climate, with scale selection varying considerably between variables and species. Contrary to our expectations mobile species did not respond at larger spatial scales, but at smaller than flightless species. Moreover, scale selection was related to the variable type, with forest structures being selected at smaller scales than variables describing the topography and forest type. Our results confirm that multi-scale models perform better than models including variables at one, a-priori selected scale, highlighting the importance of evidence-based scale-selection for species distribution models and management recommendations based thereon.
Scale selection
Variables at different spatial scales performed differently in predicting species presences. Most variables were selected at small scales with an overall median of 100 m, but with a high variance between variables and species. This variance is in line with previous studies investigating the effect of scale in Coleoptera (Økland et al. 1996; Bergman et al. 2012; Jacobsen et al. 2015), however, most of them used multispecies presence or species richness as response variables, where a greater variance can be expected compared to single species studies. For example, Holland et al. (2004) showed that species of the same family responded to forest cover at spatial scales between 20 and 2000 m. Smaller scales reflect the variability in local environmental conditions, but if chosen too small, information can get redundant. In contrast, larger scales are reducing the variability by integrating (i.e. averaging) habitat conditions of a wider surrounding which could obscure their effect (Anderson et al. 2010).
Contrary to our expectations and literature (Chust et al. 2004; Percel et al. 2019) we found a reversed pattern, that mobile species responded to variables at smaller spatial scales compared to flightless species. We propose that this pattern is explained by their ecological use of the landscape by the mobile species, as they might be able to reach and find small patches of habitat in an unsuitable forest matrix. Flightless species, in contrast, might depend on a sufficiently high density of key habitat features within a larger surrounding in order to ensure the survival of the entire population. Flightlessness is usually associated with stable habitats (Ikeda et al. 2012), where ephemeral habitat features such as deadwood have to be continuously supplied in close proximity. Richness of species with low-mobility has been shown to be highest in forests larger than 100 ha and older than 130 years, i.e. forest size and continuity determining habitat selection (Irmler et al. 2010). A similar pattern was found in butterflies, where the richness of sedentary species was positively correlated with habitat area, while there was no such correlation in mobile species (Wilcox et al. 1986). Our results suggest that the spatial scales at which habitat features were selected do not reflect individual home range size, but rather the density at which key habitat features are available in the landscape to sustain species populations. In this context, the largest of the investigated scales (1000 m) might have still been too small to encompass flying species populations, and the smallest (50 m) too large for capturing flightless species individual home ranges. Even though there is little research on effective dispersal distances in saproxylic beetles (Komonen and Müller 2018), we assume that dispersal ability and therefore range size differs strongly between flightless and mobile species. For example, males of the flying Lucanus cervus have a home range of 7585 m2 (minimum convex polygon, logNorm) or 14,487 m2 (95% kernel density estimates) (Tini et al. 2018), which would correspond to a spatial scale of 50–68 m radius. Comparatively, home range sizes of two flightless beetles in an arid environment did not exceed 700 m2 (r = 8.4 m) (Matyukhin and Gongalskii 2007). The largest analyzed scale in this study was not exceeding 1000 m, even though other studies have found the optimal scale for certain environmental variables above 1000 m (Økland et al. 1996; Holland et al. 2004; Bergman et al. 2012; Jacobsen et al. 2015; Percel et al. 2019). In our case scales above 1000 m radius would have led to overlapping windows and spatial autocorrelation.
Independent of species mobility, forest structure variables were selected at smaller scales than variables describing forest type or topography. This reflects their hierarchical clustering from fine to coarse: Forest structure variables are nested within forest type, with the latter representing available habitat, while the former rather representing resources which are selected at smaller spatial scales. Topographic variables are “independent” from the other variable types and may thus predict at different scales. Elevation is highly correlated with climatic variables such as temperature, windspeed and daily mean saturation deficit, which are expected to act at larger spatial scales than land cover (Pearson and Dawson 2003; Luoto et al. 2006). In our study both, topographic and forest type variables were predicting species occurrence probabilities at similar, larger spatial scales, since the study area is characterized by a high variation in elevation and associated topographical conditions.
In line with previous studies (e.g. Graf et al. 2005) multi-scale models performed better than single-scale models, highlighting the importance of selecting variables at their best performing scale. A-priori univariate scale selection resulted in better or equally good models (AICc delta > 2) than using all variables at the same, often arbitrarily selected, scale (Fig. 4, Table S4; Wheatley and Johnson 2009). Moreover, it has been shown that multi-scale models are especially important for mobile species, while predictions for sedentary species are less influenced if multi- or single-scaled variables are used (Meyer and Thuiller 2006).
Species habitat selection
Model performance (AUC) was higher for the flightless beetles (with fewer presence locations) than for mobile species. This result is in line with Pöyry et al. (2008) who found better predictions for less mobile butterfly species than high mobile butterflies. Although the low number of presence locations for some species like O. ferruginea and most flightless species limited the number of predictor variables and thus also model performance (Guisan et al. 2007), all models showed a sufficiently high model accuracy (AUC > 0.75), making them useful for conservation planning and downstream applications (Pearce and Ferrier 2000).
Most of the studied species preferred a mixed forest type. Echinodera hypocrita and H. imperialis were both related to mixed forests (Möller 2009; Rheinheimer and Hassler 2013; Horák and Rébl 2013), while Acalles micros and Adexius scrobipennis preferred broadleaved mixed forests. This is reflected in their prediction maps, where they are restricted to the edge of the study area, where forests are dominated by broadleaved trees. Salpingus ruficollis and Ampedus balteatus on the other hand were relying on high amounts of coniferous or coniferous mixed forest, with the former also being linked to broadleaved forest at a larger scale. Their predictions are more evenly distributed throughout the study area, as the Black Forest is mainly dominated by coniferous and mixed forests.
Forest height, a proxy for old forest, positively affected the presence of three species Adexius scrobipennis, E. hypocrita and H. imperialis. The former two flightless species are known to occur in old forests with a high degree of naturalness (Stüben 2005; Bahr and Stüben 2007), with E. hypocrita considered a relict species of ancient woodlands (Buse 2012). Even though deadwood is an ephemeral resource, old forests may continuously provide sufficient amounts not only of standing, but also of lying deadwood, which could not be assessed by remote sensing. Forest height may thus reflect the continuous availability of deadwood better than the standing deadwood variable per se. Standing deadwood was only selected into the final models of two species, P. pumilio and O. ferruginea. While P. pumilio is not classified as obligatory saproxylic, its preference to hide in leave litter and lay its eggs below woody debris or stones (Wachmann et al. 1995) may classify it as facultative saproxylic (Graf et al. 2022). Saproxylic species depend on a wide diversity of decaying deadwood and in different abiotic conditions (Seibold et al. 2016). Unfortunately, the identification of deadwood from remote sensing data does not yet provide information about deadwood quality or the stage of decay that might have improved the predictions. Moreover, lying deadwood below the canopy can neither be detected nor quantified with stereo aerial imagery. However, this information of more detailed forest attributes and deadwood is most likely necessary to make better predictions, as they represent elementary resources. As O. ferruginea is inhabiting particularly large, sun exposed standing dead trees (Möller 2009), the relationship of this species with standing deadwood can be well captured by the deadwood detection method used here (Zielewska-Büttner 2020). The presence of S. ruficollis, P. pumilio and H. imperialis was favored by high tree density, high tree volume and few gaps. All these variables reflect the species’ preference for rather dense forests (Müller et al. 2010) or indifference to sun exposure (Ranius and Jansson 2000), other than Ampedus balteatus which preferred both high tree cover and gaps.
Climate variables, especially temperature, are important drivers of the distribution of saproxylic beetles and insects in general (Bale et al. 2002), but played a minor role in our models, due to the restricted climatic range of the study area. The presence of Ampedus balteatus was best explained by low values of saturation deficit, a measure for humidity. Salpingus ruficollis, H. imperialis and E. hypocrita were found at high elevations, and P. pumilio, a cold adapted species of colline to montane altitudes (Trautner 2017), was associated with low maximum temperatures.
In this study we were focusing on the effect of different landscape elements at varying spatial scales on the presence of saproxylic beetles. However, also temporal scales of habitat and resources availability influence saproxylic beetles. The decay stage of deadwood is for example influencing the spatial scale at which saproxylic beetles respond to this resource (Jonsell et al. 2019). This is in line with theory, which predicts the colonization of ephemeral resources to be related with more mobile species, whereas more long-lasting resources to be related with less mobile species (Southwood 1977). Yet, the ecological patterns explaining the influence of temporal and spatial scales to species distributions still needs further investigations, also to derive sound management recommendations (Sverdrup-Thygeson et al. 2014).
Conclusions
Understanding species’ habitat requirements at relevant spatial scales is elementary to frame appropriate target values for forest management and conservation recommendations (Percel et al. 2019). Our study provides this information for seven obligatory and one facultative saproxylic species: Ensuring sufficient densities of resources and key habitat structures within the surrounding landscape is crucial to compensate for low dispersal ability of flightless species and to sustain viable populations. Mobile species, in contrast, are able to colonize smaller habitat patches with a high density of key structures within a larger surrounding of seemingly unsuitable forest matrix. Less mobile species may thus particularly benefit from the designation of forest reserves that locally provide high densities of key forest structural attributes within areas large enough to harbor viable populations. Retention approaches, in contrast, designed to enhance structural complexity in managed forests by sparing groups of trees or small forest patches from harvesting (Lindenmayer et al. 2012; Gustafsson et al. 2020), may be particularly beneficial for mobile species. Further investigation into effective dispersal distances of saproxylic species and their capacity to move through the landscape, would help to better understand the underlying population processes (Komonen and Müller 2018) and refine recommendations in terms of size and spacing of key structural elements at the landscape scale. In future, more saproxylic species, especially indicator species representing different ecological groups would be needed to broaden management recommendations. Investigating the requirements of single species rather than focusing on species diversity would allow to identify scale-specific response patterns in relation to species’ dispersal ability (Komonen 2008) and to derive targets for habitat management at both local and landscape scales.
Data Availability
Species data generated or analyzed during this study are provided in full within the published article. Base data and environmental variables generated for this study are not publicly available due to data ownership reasons, but can be requested at the Forest Research Institute of Baden-Wuerttemberg FVA.
References
Allouche O, Tsoar A, Kadmon R (2006) Assessing the accuracy of species distribution models: prevalence, kappa and the true skill statistic (TSS). J Appl Ecol 43:1223–1232. https://doi.org/10.1111/j.1365-2664.2006.01214.x
Anderson CD, Epperson BK, Fortin M-J et al (2010) Considering spatial and temporal scale in landscape-genetic studies of gene flow. Mol Ecol 19:3565–3575. https://doi.org/10.1111/j.1365-294X.2010.04757.x
Bahr F, Stüben PE (2007) Revision des Genus Ruteria Roudier, 1954 (Coleoptera: Curculionidae: Cryptorhynchinae). Curculio-Institute, Mönchengladbach
Bale JS, Masters GJ, Hodkinson ID et al (2002) Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Glob Change Biol 8:1–16. https://doi.org/10.1046/j.1365-2486.2002.00451.x
Bauhus J, Puettmann KJ, Kuehne C (2013) Close-to-nature forest management in Europe: does it support complexity and adaptability of forest ecosystems? Managing forests as Complex Adaptive Systems: building resilience to the challenge of global change. The Earthscan forest library, Routledge, pp 187–213
Bergman K-O, Jansson N, Claesson K et al (2012) How much and at what scale? Multiscale analyses as decision support for conservation of saproxylic oak beetles. For Ecol Manag 265:133–141. https://doi.org/10.1016/j.foreco.2011.10.030
Brunetti M, Magoga G, Iannella M et al (2019) Phylogeography and species distribution modelling of Cryptocephalus barii (Coleoptera: Chrysomelidae): is this alpine endemic species close to extinction? ZK 856:3–25. https://doi.org/10.3897/zookeys.856.32462
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Buse J (2012) Ghosts of the past”: flightless saproxylic weevils (Coleoptera: Curculionidae) are relict species in ancient woodlands. J Insect Conserv 16:93–102. https://doi.org/10.1007/s10841-011-9396-5
Chust G, Pretus JLl, Ducrot D, Ventura D (2004) Scale dependency of insect assemblages in response to landscape pattern. Landscape Ecol 19:41–57. https://doi.org/10.1023/B:LAND.0000018368.99833.f2
Crawford PHC, Hoagland BW (2010) Using species distribution models to guide conservation at the state level: the endangered american burying beetle (Nicrophorus americanus) in Oklahoma. J Insect Conserv 14:511–521. https://doi.org/10.1007/s10841-010-9280-8
Della Rocca F, Bogliani G, Milanesi P (2017) Patterns of distribution and landscape connectivity of the stag beetle in a human-dominated landscape. NC 19:19–37. https://doi.org/10.3897/natureconservation.19.12457
Dietrich H, Wolf T, Kawohl T et al (2019) Ann For Sci 76. https://doi.org/10.1007/s13595-018-0788-5. Temporal and spatial high-resolution climate data from 1961 to 2100 for the German National Forest Inventory (NFI)
Dittrich A, Roilo S, Sonnenschein R et al (2019) Modelling distributions of Rove Beetles in mountainous areas using Remote Sensing Data. Remote Sens 12:80. https://doi.org/10.3390/rs12010080
Eckerter T, Braunisch V, Pufal G, Klein AM (2022) Small clear-cuts in managed forests support trap-nesting bees, wasps and their parasitoids. For Ecol Manag 509:120076. https://doi.org/10.1016/j.foreco.2022.120076
Elith J, Graham H, Anderson CP R, et al (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151. https://doi.org/10.1111/j.2006.0906-7590.04596.x
Evans JS, Murphy MA, Ram K (2021) Spatial Analysis and Modelling Utilities
Eyre MD, Rushton SP, Luff ML, Telfer MG (2005) Investigating the relationships between the distribution of british ground beetle species (Coleoptera, Carabidae) and temperature, precipitation and altitude: british ground beetles, temperature, precipitation and altitude. J Biogeogr 32:973–983. https://doi.org/10.1111/j.1365-2699.2005.01258.x
FAO (2010) Globa Forest resource Assessment 2010. Rome, Italy
Fielding AH, Bell JF (1997) A review of methods for the assessment of prediction errors in conservation presence/absence models. Envir Conserv 24:38–49. https://doi.org/10.1017/S0376892997000088
Freude H, Harde KW, Lohse (1969) Die Käfer Mitteleuropas. Goecke & Evers Verlag, Krefeld
Freude H, Harde KW, Lohse (1979) Die Käfer Mitteleuropas. Goecke & Evers Verlag, Krefeld
Freude H, Harde KW, Lohse (1983) Die Käfer Mitteleuropas. Goecke & Evers Verlag, Krefeld
Ganz S, Adler P, Kändler G (2020) Forests 11:1322. https://doi.org/10.3390/f11121322. Forest Cover Mapping Based on a Combination of Aerial Images and Sentinel-2 Satellite Data Compared to National Forest Inventory Data
Gao T, Nielsen AB, Hedblom M (2015) Reviewing the strength of evidence of biodiversity indicators for forest ecosystems in Europe. Ecol Ind 57:420–434. https://doi.org/10.1016/j.ecolind.2015.05.028
Gehring TM, Swihart RK (2003) Body size, niche breadth, and ecologically scaled responses to habitat fragmentation: mammalian predators in an agricultural landscape. Biol Conserv 109:283–295. https://doi.org/10.1016/S0006-3207(02)00156-8
Graf RF, Bollmann K, Suter W, Bugmann H (2005) The importance of spatial scale in Habitat Models: Capercaillie in the Swiss Alps. Landsc Ecol 20:703–717. https://doi.org/10.1007/s10980-005-0063-7
Graf M, Seibold S, Gossner MM et al (2022) Coverage based diversity estimates of facultative saproxylic species highlight the importance of deadwood for biodiversity. For Ecol Manag 517:120275. https://doi.org/10.1016/j.foreco.2022.120275
Guisan A, Graham CH, Elith J et al (2007) Sensitivity of predictive species distribution models to change in grain size. Divers Distrib 13:332–340. https://doi.org/10.1111/j.1472-4642.2007.00342.x
Gustafsson L, Bauhus J, Asbeck T et al (2020) Retention as an integrated biodiversity conservation approach for continuous-cover forestry in Europe. Ambio 49:85–97. https://doi.org/10.1007/s13280-019-01190-1
Harrell FE, Lee KL, Mark DB (1996) Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Statist Med 15:361–387. https://doi.org/10.1002/(SICI)1097-0258(19960229)15:4<361::AID-SIM168>3.0.CO;2-4
He KS, Bradley BA, Cord AF et al (2015) Will remote sensing shape the next generation of species distribution models? Remote Sens Ecol Conserv 1:4–18. https://doi.org/10.1002/rse2.7
Hijmans RJ, Phillips S, Leathwick J, Elith J (2020) dismo: Species Distribution Modeling
Hijmans RJ, van Etten J, Summer M et al (2022) Package ‘raster’
Hjältén J, Stenbacka F, Pettersson RB et al (2012) Micro and Macro-Habitat Associations in Saproxylic Beetles: implications for Biodiversity Management. PLoS ONE 7:e41100. https://doi.org/10.1371/journal.pone.0041100
Hof AR, Svahlin A (2016) The potential effect of climate change on the geographical distribution of insect pest species in the swedish boreal forest. Scand J For Res 31:29–39. https://doi.org/10.1080/02827581.2015.1052751
Holland JD, Bert DG, Fahrig L (2004) Determining the spatial scale of Species’ response to Habitat. Bioscience 54:227. https://doi.org/10.1641/0006-3568(2004)054[0227:DTSSOS]2.0.CO;2
Horák J, Rébl K (2013) The species richness of click beetles in ancient pasture woodland benefits from a high level of sun exposure. J Insect Conserv 17:307–318. https://doi.org/10.1007/s10841-012-9511-2
Ikeda H, Nishikawa M, Sota T (2012) Loss of flight promotes beetle diversification. Nat Commun 3:648. https://doi.org/10.1038/ncomms1659
Irmler U, Arp H, Nötzold R (2010) Species richness of saproxylic beetles in woodlands is affected by dispersion ability of species, age and stand size. J Insect Conserv 14:227–235. https://doi.org/10.1007/s10841-009-9249-7
Jacobsen RM, Sverdrup-Thygeson A, Birkemoe T (2015) Scale-specific responses of saproxylic beetles: combining dead wood surveys with data from satellite imagery. J Insect Conserv 19:1053–1062. https://doi.org/10.1007/s10841-015-9821-2
Jiménez-Valverde A, Ortuño VM, Lobo JM (2007) Exploring the distribution of Sterocorax Ortuño, 1990 (Coleoptera, Carabidae) species in the Iberian Peninsula: distribution of Sterocorax species in the Iberian Peninsula. J Biogeogr 34:1426–1438. https://doi.org/10.1111/j.1365-2699.2007.01702.x
Jonsell M, Abrahamsson M, Widenfalk L, Lindbladh M (2019) Increasing influence of the surrounding landscape on saproxylic beetle communities over 10 years succession in dead wood. For Ecol Manag 440:267–284. https://doi.org/10.1016/j.foreco.2019.02.021
Judas M, Dornieden K, Strothmann U (2002) Distribution patterns of carabid beetle species at the landscape-level. J Biogeogr 29:491–508. https://doi.org/10.1046/j.1365-2699.2002.00697.x
Kärvemo S, Van Boeckel TP, Gilbert M et al (2014) Large-scale risk mapping of an eruptive bark beetle – importance of forest susceptibility and beetle pressure. For Ecol Manag 318:158–166. https://doi.org/10.1016/j.foreco.2014.01.025
Kenis M, Wermelinger B, Grégoire J-C (2004) Research on parasitoids and predators of scolotidae - a review. Bark and wood boring insects in living trees in Europe, a synthesis. Kluwer Academic Publishers, pp 237–290
Knuff AK, Winiger N, Klein A et al (2019) Optimizing sampling of flying insects using a modified window trap. Methods Ecol Evol 10:1820–1825. https://doi.org/10.1111/2041-210X.13258
Koch K (1992) Die Käfer Mitteleuropas Ökologie. Goecke & Evers Verlag, Krefeld
Komonen A (2008) Colonization experiment of fungivorous beetles (Ciidae) in a lake-island system. 5
Komonen A, Müller J (2018) Dispersal ecology of deadwood organisms and connectivity conservation. Conserv Biol 32:535–545. https://doi.org/10.1111/cobi.13087
Kraut A, Liira J, Lõhmus A (2016) Beyond a minimum substrate supply: sustaining saproxylic beetles in semi-natural forest management. For Ecol Manag 360:9–19. https://doi.org/10.1016/j.foreco.2015.10.016
Lachat T, Wermelinger B, Gossner MM et al (2012) Saproxylic beetles as indicator species for dead-wood amount and temperature in european beech forests. Ecol Ind 23:323–331. https://doi.org/10.1016/j.ecolind.2012.04.013
Landesamt für Geoinformation und Landentwicklung Baden-Württemberg (LGL). (2018) Geobasisdaten. www.lgl-bw.deaz.:2851. Accessed 15 Aug 2019
Levin SA (1992) The Problem of Pattern and Scale in Ecology: the Robert H. MacArthur Award Lecture Ecology 73:1943–1967. https://doi.org/10.2307/1941447
Lindenmayer DB, Franklin JF (2002) Conserving Forest Biodiversity: a Comprehensive Multiscaled Approach. Island Press, Washington, D.C.
Lindenmayer DB, Franklin JF, Lõhmus A et al (2012) A major shift to the retention approach for forestry can help resolve some global forest sustainability issues: Retention forestry for sustainable forests. Conserv Lett 5:421–431. https://doi.org/10.1111/j.1755-263X.2012.00257.x
Luoto M, Virkkala R, Heikkinen RK (2006) The role of land cover in bioclimatic models depends on spatial resolution. Global Ecol Biogeography 0:061120101210017–. https://doi.org/10.1111/j.1466-822X.2006.00262.x
Matyukhin AV, Gongalskii KB (2007) Home range size in two darkling beetle species (Coleoptera, Tenebrionidae) from Southern Kazakhstan. Entmol Rev 87:1232–1237. https://doi.org/10.1134/S0013873807090126
McGarigal K, Wan HY, Zeller KA et al (2016) Multi-scale habitat selection modeling: a review and outlook. Landsc Ecol 31:1161–1175. https://doi.org/10.1007/s10980-016-0374-x
Meyer CB, Thuiller W (2006) Accuracy of resource selection functions across spatial scales. Divers Distrib 12:288–297. https://doi.org/10.1111/j.1366-9516.2006.00241.x
Möller G (2009) Struktur-und Substratbindung holzbewohnender Insekten, Schwerpunkt Coleoptera-Käfer. PhD Thesis
Müller J, Engel H, Blaschke M (2007) Assemblages of wood-inhabiting fungi related to silvicultural management intensity in beech forests in southern Germany. Eur J For Res 126:513–527. https://doi.org/10.1007/s10342-007-0173-7
Müller J, Noss RF, Bussler H, Brandl R (2010) Learning from a “benign neglect strategy” in a national park: response of saproxylic beetles to dead wood accumulation. Biol Conserv 143:2559–2569. https://doi.org/10.1016/j.biocon.2010.06.024
Müller J, Wende B, Strobl C et al (2015) Forest management and regional tree composition drive the host preference of saproxylic beetle communities. J Appl Ecol 52:753–762. https://doi.org/10.1111/1365-2664.12421
Økland B, Bakke A, H\a agvar S, Kvamme T (1996) What factors influence the diversity of saproxylic beetles? A multiscaled study from a spruce forest in southern Norway. Biodivers Conserv 5:75–100
Paillet Y, BergèS L, HjäLtéN J et al (2010) Biodiversity differences between Managed and unmanaged forests: Meta-Analysis of Species Richness in Europe. Conserv Biol 24:101–112. https://doi.org/10.1111/j.1523-1739.2009.01399.x
Pearce J, Ferrier S (2000) Evaluating the predictive performance of habitat models developed using logistic regression. Ecol Model 133:225–245. https://doi.org/10.1016/S0304-3800(00)00322-7
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Global Ecol Biogeogr 12:361–371
Percel G, Laroche F, Bouget C (2019) The scale of saproxylic beetles response to landscape structure depends on their habitat stability. Landsc Ecol 34:1905–1918. https://doi.org/10.1007/s10980-019-00857-0
Phillips SJ, Dudík M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31:161–175. https://doi.org/10.1111/j.0906-7590.2008.5203.x
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Pinheiro J, Bates D, DebRoy S, Sarkar D (2020) nlme: Linear and Nonlinear Mixed Effects Models
Pöyry J, Luoto M, Heikkinen RK, Saarinen K (2008) Species traits are associated with the quality of bioclimatic models. Glob Ecol Biogeogr 17:403–414. https://doi.org/10.1111/j.1466-8238.2007.00373.x
Ranius T, Jansson N (2000) The influence of forest regrowth, original canopy cover and tree size on saproxylic beetles associated with old oaks. Biol Conserv 95:85–94
Reddy CS, Kurian A, Srivastava G et al (2021) Remote sensing enabled essential biodiversity variables for biodiversity assessment and monitoring: technological advancement and potentials. Biodivers Conserv 30:1–14. https://doi.org/10.1007/s10531-020-02073-8
Rheinheimer J, Hassler M (2013) Die Rüsselkäfer Baden-Württembergs, 2nd edn. Verlag Regionalkultur, Ubstadt-Weiher
Rothermel M, Wenzel K, Fritsch D, Haala N (2012) SURE: photogrammetric surface reconstruction from imagery. In: LC3D Workshop. Berlin, Germany, p 9
Schmidl VJ, Bussler H (2004) Ökologische Gilden xylobionter Käfer Deutschlands. Naturschutz und Landschaftsplanung 17
Schumacher J, Rattay M, Kirchhöfer M et al (2019) Combination of Multi-Temporal Sentinel 2 images and aerial image based canopy height models for timber volume modelling. Forests 10:746. https://doi.org/10.3390/f10090746
Seibold S, Brandl R, Buse J et al (2015) Association of extinction risk of saproxylic beetles with ecological degradation of forests in Europe. Conserv Biol 29:382–390. https://doi.org/10.1111/cobi.12427
Seibold S, Bässler C, Brandl R et al (2016) Microclimate and habitat heterogeneity as the major drivers of beetle diversity in dead wood. J Appl Ecol 53:934–943. https://doi.org/10.1111/1365-2664.12607
Seibold S, Gossner MM, Simons NK et al (2019) Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574:671–674. https://doi.org/10.1038/s41586-019-1684-3
Silva DP, Aguiar AG, Simião-Ferreira J (2016) Assessing the distribution and conservation status of a long-horned beetle with species distribution models. J Insect Conserv 20:611–620. https://doi.org/10.1007/s10841-016-9892-8
Southwood TRE (1977) Habitat, the Templet for ecological strategies? J Anim Ecol 46:336. https://doi.org/10.2307/3817
Speight MCD (1989) Saproxylic invertebrates and their conservation. Nat Environ Ser 42:1–79
Steffan-Dewenter I, Münzenberg U, Bürger C et al (2002) Scale-dependent effects of landscape context on three pollinator guilds. Ecology 83:1421–1432. https://doi.org/10.1890/0012-9658(2002)083[1421:SDEOLC]2.0.CO;2
Storch I, Penner J, Asbeck T et al (2020) Evaluating the effectiveness of retention forestry to enhance biodiversity in production forests of Central Europe using an interdisciplinary, multi-scale approach. Ecol Evol. https://doi.org/10.1002/ece3.6003
Stüben PE (2005) Isoliertes Vorkommen einer Acalles micros-Population in einem Buchenwald des Niederbergischen Landes (Curculionidae: Cryptorhynchinae). Curculio-Institute, Mönchengladbach
Sverdrup-Thygeson A, Gustafsson L, Kouki J (2014) Spatial and temporal scales relevant for conservation of dead-wood associated species: current status and perspectives. Biodivers Conserv 23:513–535. https://doi.org/10.1007/s10531-014-0628-3
Tini M, Bardiani M, Chiari S et al (2018) Use of space and dispersal ability of a flagship saproxylic insect: a telemetric study of the stag beetle (Lucanus cervus) in a relict lowland forest. Insect Conserv Divers 11:116–129. https://doi.org/10.1111/icad.12260
Trautner J (2017) Die Laufkäfer Baden-Württembergs. Eugen Ulmer KG, Stuttgart
Turner JA, Babcock RC, Kendrick GA, Hovey RK (2019) How does spatial resolution affect model performance? A case for ensemble approaches for marine benthic mesophotic communities. J Biogeogr 46:1249–1259. https://doi.org/10.1111/jbi.13581
Urban P (2019) Ein zoogeographisch und ökologisch beachtenswerter Käferfund im Eggegebirge in Ostwestfalen: Peltis ferruginea (LINNAEUS, 1758)(Coleoptera, Peltidae). Mitt ArbGem westfäl Entomol 35:15–18
Vignali S, Barras AG, Arlettaz R, Braunisch V (2020) SDMtune: an R package to tune and evaluate species distribution models. Ecol Evol 10:11488–11506. https://doi.org/10.1002/ece3.6786
Wachmann E, Platen R, Barndt D (1995) Laufkäfer: Beobachtung, Lebensweise. Naturbuch Verlag, Augsburg
Wheatley M, Johnson C (2009) Factors limiting our understanding of ecological scale. Ecol Complex 6:150–159. https://doi.org/10.1016/j.ecocom.2008.10.011
Wilcox BA, Murphy DD, Ehrlich PR, Austin GT (1986) Insular biogeography of the montane butterfly faunas in the Great Basin: comparison with birds and mammals. Oecologia 69:188–194. https://doi.org/10.1007/BF00377620
Zielewska-Büttner K, Adler P, Petersen M, Braunisch V (2016) Parameters influencing forest gap detection using canopy height models derived from Stereo Aerial Imagery. DGPF, OVG, SGPF, Bern
Zielewska-Büttner K, Adler P, Kolbe S et al (2020) Detection of Standing Deadwood from Aerial Imagery Products: Two Methods for Addressing the Bare Ground Misclassification Issue. forests 11:1–26
Acknowledgements
We are thankful for the help of numerous technical and student assistants with field- and lab work. Thanks to Christoph Germann (Natural History Museum Basel) and Werner Marggi (Thun) for the identification of the Curculionidae and Carabidae specimens. We are grateful to the State’s Forest Administration of Baden-Württemberg (ForstBW) for enabling and facilitating field work. In addition we thank two anonymous reviewers for insightful comments on an earlier version of this manuscript.
Funding
This study was funded by the German Research Foundation within the Research Training Group ConFoBi (grant number GRK 2123/1 TPX) and the “Special Program for Promoting Biological Diversity” (Sonderprogramm zur Stärkung der Biologischen Vielfalt) of the Ministry of Food, Rural Areas and Consumer Protection Baden-Württemberg.
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
N.W., V.B. and G.S. conceptualized the study. N.W., A-L.H., S.G. and K.Z.-B. collected and prepared data. N.W. and A-L.H. conducted the analysis. N.W. wrote the manuscript with input of all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Nigel Stork.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Winiger, N., Hendel, AL., Ganz, S. et al. Saproxylic beetles respond to habitat variables at different spatial scales depending on variable type and species’ mobility: the need for multi-scale forest structure management. Biodivers Conserv 32, 3355–3377 (2023). https://doi.org/10.1007/s10531-023-02663-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02663-2